Product Formulation and Regulatory Compliance: A Pre-Launch Guide for Researchers
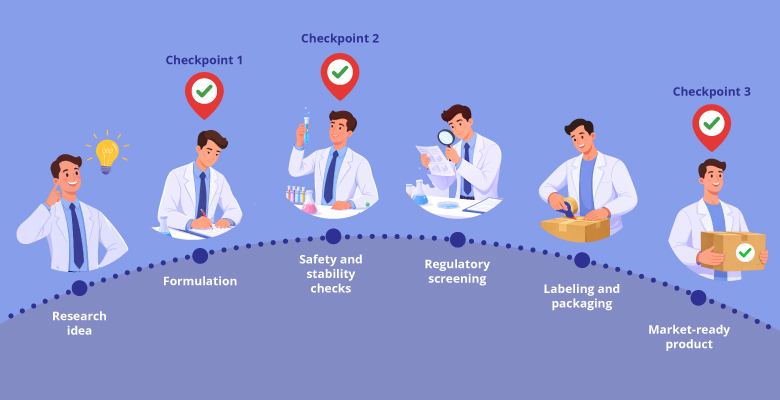
Every innovative product begins with a compelling idea—yet only a fraction of those ideas successfully reaches the market. The limiting factor is rarely weak science or insufficient data. More often, it is a failure to anticipate regulatory requirements until it is too late. When compliance gaps surface near the commercialization stage, the consequences are steep: costly reformulation, additional testing, and significant delays that could have been avoided with better early-stage planning.
This challenge is only growing. Across pharmaceuticals, food, advanced materials, and cosmetics, researchers now have access to powerful tools and novel ingredients that accelerate formulation and prototyping. At the same time, regulatory frameworks governing safety, labeling, and efficacy have become more rigorous and increasingly region-specific. A formulation that performs well under controlled conditions may still fall short when evaluated against the standards set by bodies like the U.S. Food and Drug Administration (USFDA) or the European Commission. Safety, stability, ingredient restrictions, and labeling compliance all influence regulatory approval. The core problem is that too many researchers treat formulation and regulatory compliance as separate processes—when in fact, they are deeply interdependent.
Formulation as an Early Regulatory Decision
During the formulation stage, researchers often concentrate on whether a product “works”—its efficacy, stability, and user experience. However, formulation is also the first, and perhaps the most critical, regulatory checkpoint. Decisions around ingredient selection, concentration levels, processing methods, delivery systems, and even packaging all play a direct role in determining whether a product will meet compliance requirements across its intended target markets.
The USFDA places the burden of product safety on manufacturers even when pre-market approval is not required. The European Commission enforces strict rules on ingredient safety, permissible concentrations, and labeling under EU Regulation. Formulation, therefore, is not just about performance—it is about legal permissibility.
Consider a cosmetic product: a formulation may excel in sensory properties and demonstrated efficacy, yet inclusion of a restricted substance—or use of a permitted substance above allowable concentration limits—renders it non-compliant. The same logic applies to functional foods and nutraceuticals, where an ingredient may show clear benefits in studies but fail regulatory thresholds for dosage, additives, or claims.
Early decisions on preservative systems, excipients, and active ingredient levels can ultimately determine whether a product is approvable, scalable, and market-ready. Formulation should therefore be understood not merely as the phase where an idea is validated, but as the foundational step toward regulatory compliance and successful commercialization.
Common Pitfalls in Product Commercialization
Despite advances in formulation science and increased access to knowledge, several pitfalls continue to delay or derail product commercialization. All of them share a common thread: being reactive rather than proactive about compliance.
- Treating regulatory compliance as a late-stage activity: When regulatory requirements are only evaluated at launch, discovering that an ingredient is restricted or that additional safety data is needed can trigger significant rework—delaying timelines and inflating costs.
- Underestimating regional regulatory differences: A product developed for one market does not automatically meet the requirements of another. The European Commission mandates safety assessments, toxicological data, and detailed technical documentation before market entry. The USFDA, by contrast, follows a largely post-market enforcement model for categories like cosmetics, placing responsibility squarely on the manufacturer. In India, the Central Drugs Standard Control Organisation continues to evolve its standards, with growing emphasis on safety and labeling. Without early awareness of these differences, companies may end up maintaining multiple formulations across markets, thereby complicating manufacturing and supply chains considerably. Global commercialization requires regulatory foresight, not post-hoc adaptation.
- Over-reliance on “natural” or “clean” positioning: Consumer demand for natural products is real, but regulatory authorities evaluate safety based on scientific evidence, not marketing narratives. Natural ingredients can still present risks related to allergens, variability, or contamination, and must be assessed with the same rigor as synthetic compounds.
- Inadequate testing and documentation: Regulatory frameworks often require detailed records of formulation, testing protocols, safety assessments, and manufacturing processes. Missing or incomplete documentation can lead to compliance issues even if the product itself is safe.
- Overlooking the importance of claims substantiation: Marketing claims that are not adequately supported by data can attract scrutiny from bodies such as the Federal Trade Commission, leading to penalties or reputational damage.
Integrating Compliance into the Product Lifecycle
Integrating compliance into product development requires attention to several critical factors. The most critical of these is perhaps ingredient selection. While functionality and performance remain essential, compliance considerations are equally critical. Bodies such as the Scientific Committee on Consumer Safety evaluate ingredients based on toxicological profiles, exposure levels, and long-term safety. Researchers must therefore assess not only whether an ingredient works, but whether it is permitted, within allowable concentration limits, and appropriately classified.
Beyond ingredient selection, products must demonstrate safety, stability, and consistency. This involves stability testing under stress conditions, microbial assessments, toxicological evaluations, and shelf-life validation. In regulated markets, this data is compiled into formal safety dossiers. Testing should not be approached as a regulatory hurdle; it is central to product integrity and risk management.
Equally critical is how products are communicated. Claims related to efficacy, safety, or sustainability must be evidence-based and not misleading, as emphasized by the Federal Trade Commission. For example, “anti-aging” or “eco-friendly” claims require substantiation through validated studies or lifecycle data.
Finally, labeling, packaging, and traceability form the product’s regulatory interface. Accurate ingredient disclosure, safety warnings, and batch traceability are essential for compliance and consumer safety. Packaging must also ensure stability and prevent contamination.
Navigating all of this often requires expertise that a single development team may not have in-house. For researchers and early-stage developers, this can create real bottlenecks — particularly when working across complex regulatory environments or specialized testing requirements. External collaboration can help bridge these gaps. Platforms like Kolabtree, for instance, connect researchers with formulation scientists, regulatory experts, and industry professionals on a flexible basis, providing targeted support where it is most needed.
The Road Ahead
Undoubtedly, researchers aiming for commercialization must integrate regulatory thinking early, ideally as soon as preliminary results indicate commercial potential. As industries evolve, regulatory frameworks are becoming more sophisticated, with greater demand for transparency, safety, and sustainability.
Product commercialization can no longer follow a linear path from formulation to market. It must be an integrated process, one where scientific innovation and regulatory expectations advance in parallel. Success will be determined not only by how well a product performs, but by how effectively it navigates compliance, scalability, and consumer trust from the outset.
References
- Why Innovations Fail to Reach the Market: The Tragic Reality of Academic Research. Conlis Global. https://conlisglobal.com/why-innovations-fail-to-reach-the-market-the-tragic-reality-of-academic-research/ Accessed on 27th March 2026.
- FDA Regulatory Information. https://www.fda.gov/regulatory-information Accessed on 27th March 2026.
- European Commission. https://commission.europa.eu/index_en Accessed on 27th March 2026.
- Sengupta et al. Current regulatory requirements and practical approaches for stability analysis of pharmaceutical products: A comprehensive review. International Journal of Pharmaceutics. 2018;543:328–344. https://doi.org/10.1016/j.ijpharm.2018.04.007
- The importance of the Product Information File (PIF). Biorius. https://biorius.com/cosmetic-news/what-is-a-pif-product-information-file/ Accessed on 27th March 2026.
- How FDA Approves Drugs and Regulates Their Safety and Effectiveness. EveryCRSreport.com. https://www.everycrsreport.com/reports/R41983. Accessed on 27th March 2026.
- FDA Regulations for Dietary Supplement Manufacturing Explained. Swasthum. https://swasthum.com/fda-regulations-for-dietary-supplement-manufacturing/ Accessed on 27th March 2026.
- Mandatory food information. European Commission. https://food.ec.europa.eu/food-safety/labelling-and-nutrition/food-information-consumers-legislation/mandatory-food-information_en Accessed on 27th March 2026.
- Central Drugs Standard Control Organization. https://cdsco.gov.in/opencms/opencms/en/Home/ Accessed on 27th March 2026.
- News from CRIS: What’s the Risk? Synthetic vs. Natural Chemical Ingredients. https://iit.msu.edu/news/2025-4-7-CRIS-trending-GRAS-loophole Accessed on 27th March 2026.
- Federal Trade Commission. https://www.ftc.gov Accessed on 27th March 2026.
- Scientific Committee on Consumer Safety (SCCS). https://health.ec.europa.eu/scientific-committees/scientific-committee-consumer-safety-sccs_en Accessed on 27th March 2026.
- Kolabtree. https://www.kolabtree.com/how-it-works Accessed on 27th March 2026.








